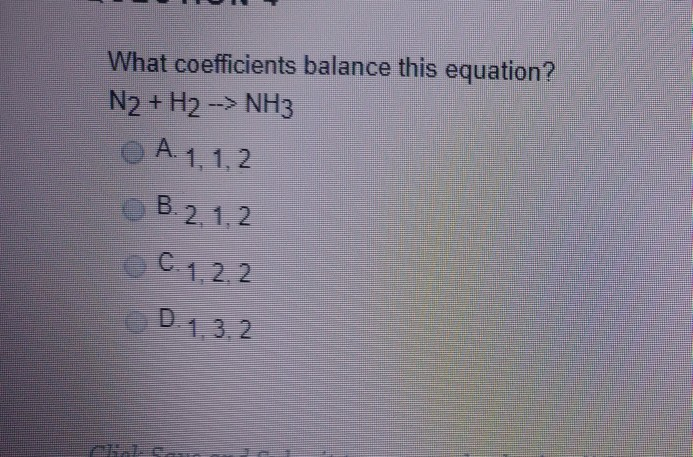
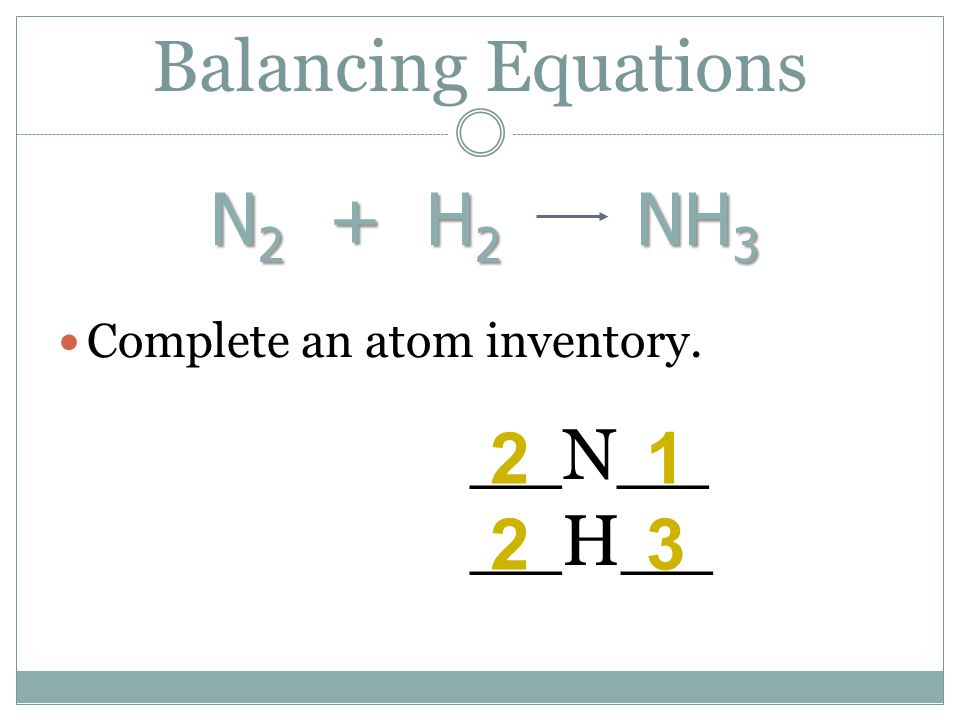
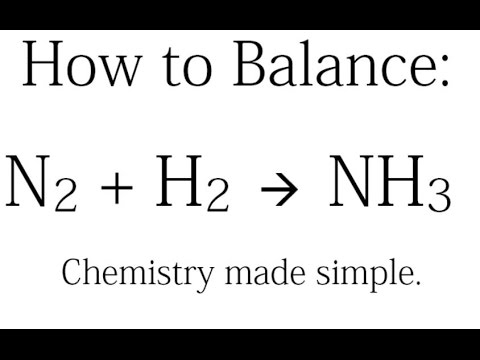
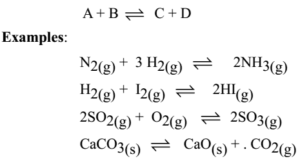Oxidation Number method. N2+H2=NH3. Balance the chemical equation by oxidation Number method. - YouTube

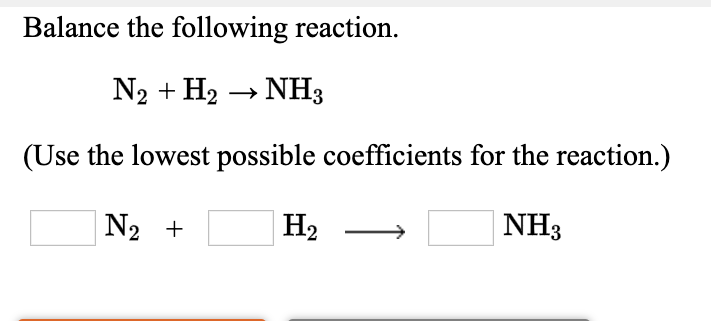
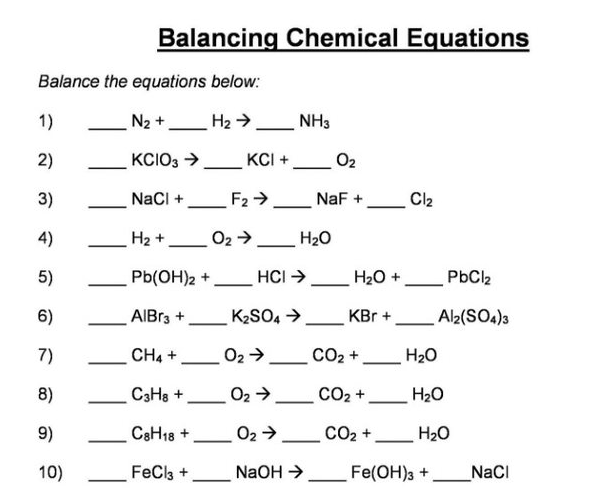
The chemical equation below shows the formation of ammonia from nitrogen and hydrogen gases. N2 + H2 - Brainly.com

N2 +H2 =NH3 Balanced Equation - n2 + h2 → nh3 balance||Nitrogen+Hydrogen=Ammonia Balanced Equation - YouTube
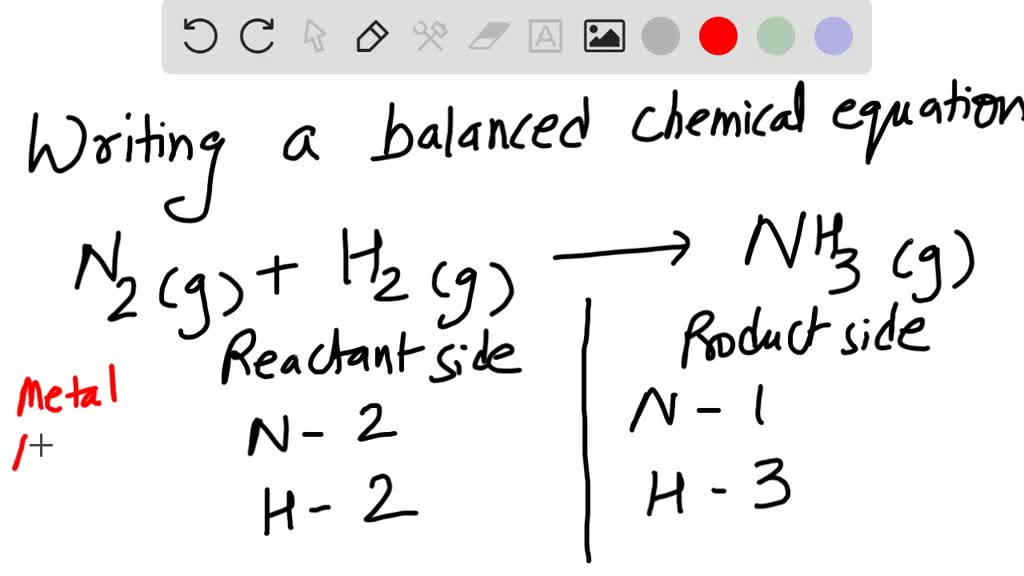
SOLVED:Write a balanced chemical equation for the production of ammonia, NH3(g), from N2(g) and H2(g)