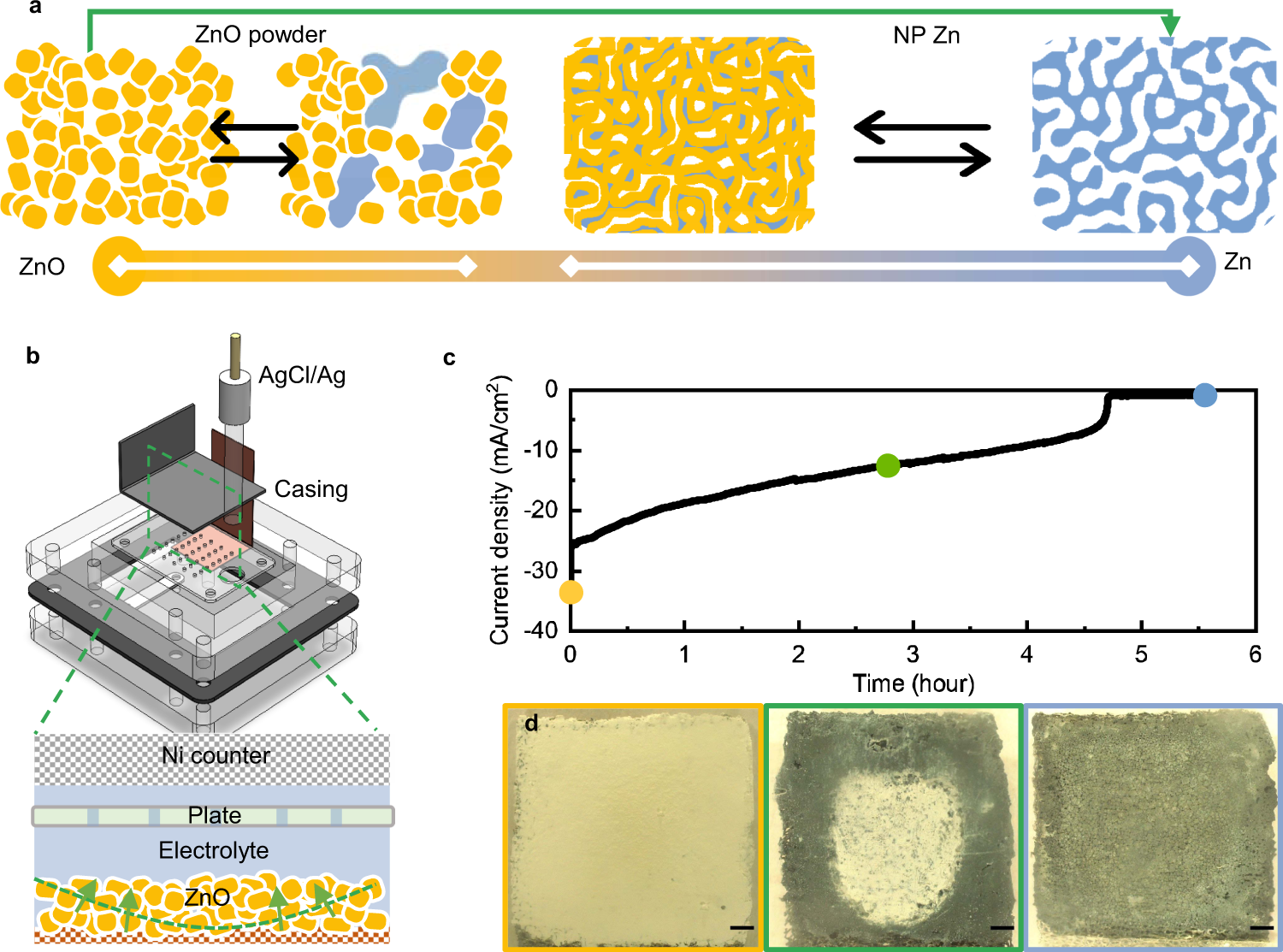
Phase-transition tailored nanoporous zinc metal electrodes for rechargeable alkaline zinc-nickel oxide hydroxide and zinc-air batteries | Nature Communications
VYSOKÉ UČENÍ TECHNICKÉ V BRNĚ MODIFIKACE MATERIÁLŮ PRO KLADNÉ ELEKTRODY LITHNO-IONTOVÝCH AKUMULÁTORŮ

PDF) Characterization of a zinc-nickel alloy coating obtained from an electrolytic bath produced with spent batteries as raw materials

Breaking the 2 V Barrier in Aqueous Zinc Chemistry: Creating 2.45 and 2.8 V MnO2–Zn Aqueous Batteries | ACS Energy Letters

Root Reason for the Failure of a Practical Zn–Ni Battery: Shape Changing Caused by Uneven Current Distribution and Zn Dissolution | ACS Applied Materials & Interfaces

Root Reason for the Failure of a Practical Zn–Ni Battery: Shape Changing Caused by Uneven Current Distribution and Zn Dissolution | ACS Applied Materials & Interfaces
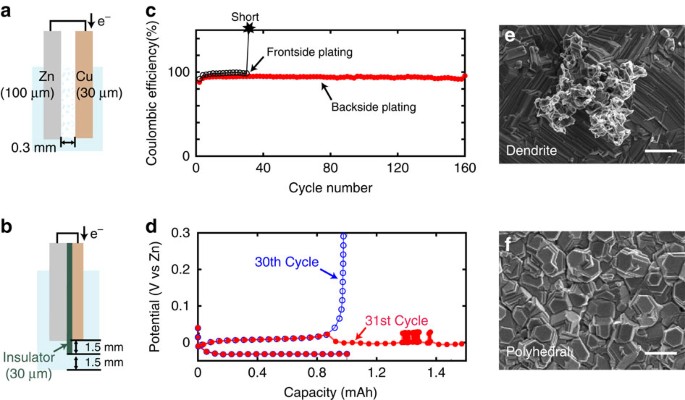
Avoiding short circuits from zinc metal dendrites in anode by backside-plating configuration | Nature Communications

Avoiding short circuits from zinc metal dendrites in anode by backside-plating configuration | Nature Communications
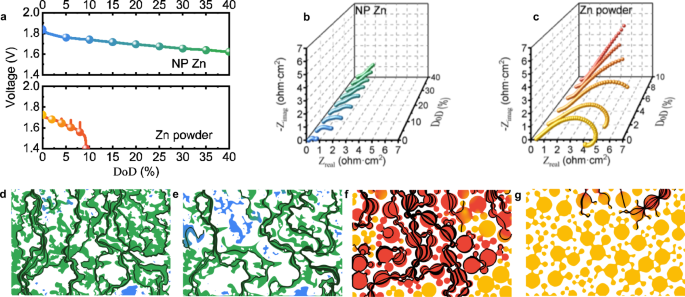
Phase-transition tailored nanoporous zinc metal electrodes for rechargeable alkaline zinc-nickel oxide hydroxide and zinc-air batteries | Nature Communications

Root Reason for the Failure of a Practical Zn–Ni Battery: Shape Changing Caused by Uneven Current Distribution and Zn Dissolution | ACS Applied Materials & Interfaces
VYSOKÉ UČENÍ TECHNICKÉ V BRNĚ MODIFIKACE MATERIÁLŮ PRO KLADNÉ ELEKTRODY LITHNO-IONTOVÝCH AKUMULÁTORŮ

Root Reason for the Failure of a Practical Zn–Ni Battery: Shape Changing Caused by Uneven Current Distribution and Zn Dissolution | ACS Applied Materials & Interfaces

Root Reason for the Failure of a Practical Zn–Ni Battery: Shape Changing Caused by Uneven Current Distribution and Zn Dissolution | ACS Applied Materials & Interfaces
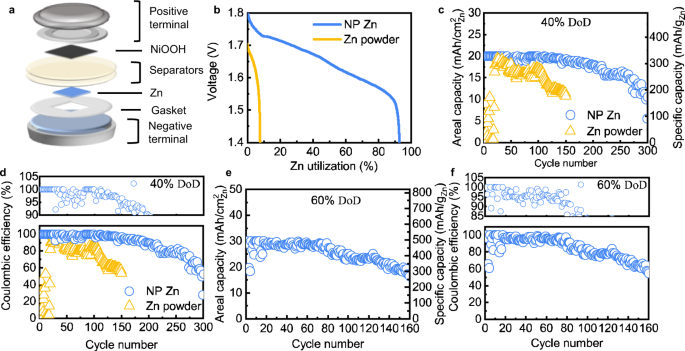
Phase-transition tailored nanoporous zinc metal electrodes for rechargeable alkaline zinc-nickel oxide hydroxide and zinc-air batteries | Nature Communications

Understanding of the electrochemical behaviors of aqueous zinc–manganese batteries: Reaction processes and failure mechanisms - ScienceDirect